
Thus, Ca 2+ uptake would not normally be expected to lower the membrane potential, at least for a sustained period, or to such an extent that it would inhibit ATP synthesis. This phenomenon can be observed clearly in isolated mitochondria at supraphysiological concentrations of extramitochondrial Ca 2+ however, under physiological conditions of Ca 2+ uptake, a change in ΔΨ m has not been detected in isolated cardiomyocytes using fluorescent indicators. Ca 2+ uptake is respiration-dependent, thus high rates of uptake can effectively lower the mitochondrial membrane potential (ΔΨ m) (reviewed in ). Average m is controlled by the activities of the MCU and the efflux pathway, the mitochondrial Na +/Ca 2+ exchanger (mNCX). Increases in c are relayed to the mitochondria by the mitochondrial Ca 2+ uniporter (MCU), resulting in activation of mitochondrial dehydrogenases and stimulation of ATP synthesis. However, recent work has very likely identified the proteins responsible for both the Ca 2+ influx and Ca 2+ efflux pathways in mitochondria (see below). ģ. Mitochondrial Ca 2+ transport: roles of the mitochondrial uniporter MCU and the Na +/Ca 2+ exchanger, mNCX/NCLXĪn impediment to progress in this field has been that the mitochondrial Ca 2+ transport proteins were not purified or cloned, and that use of the inhibitors of these pathways is problematic in living cells. Subsequent refinements of these approaches, including the use of GFP-based probes, highlighted the importance of close contacts between the endoplasmic reticulum and mitochondria in the uptake of Ca 2+ by the latter and demonstrated that mitochondrial Ca 2+ increases stably elevate mitochondrial.
ATP PRODUCTION FREE
≤100 nM) and that the free concentrations of the ion increased within mitochondria in response to Ca 2+-mobilising agonists. to measurements at the level of single cells, this approach provided evidence that basal intramitochondrial Ca 2+ levels were indeed low (similar to or lower than those in the cytosol, i.e. These experiments used recombinant expression of the Ca 2+-sensitive photoprotein aequorin, targeted to the mitochondrial matrix by in-frame fusion with the signal peptide of cytochrome c subunit VIII. ) upon which the above model was based were subsequently complemented and reinforced in the 1990s by highly selective measurements of intramitochondrial free Ca 2+ in living cells by Rizzuto, Pozzan, Rutter and others. Export of mitochondrial ATP in exchange for ADP was anticipated then to meet an increased ATP demand to fuel energy-requiring processes in the cytosol such as ion pumping, contraction, exocytosis etc. The consequent activation of oxidative metabolism would then provide an increased supply of reducing equivalents to drive respiratory chain activity and ATP synthesis. Thus, based on the properties of a group of key intramitochondrial dehydrogenases (later characterised in some detail – see the following sections), it was proposed that the uptake of Ca 2+ ions from the cytosol increased m from ∼0.1 to 10 μM or more. Studies in the 1970s and 80s by Denton and McCormack as well as Hansford and Castro and Crompton indicated instead that under basal conditions total mitochondrial Ca 2+ content was low and that increases in cytosolic free Ca 2+ ( c) in response to extrinsic agents (nutrients, hormones, neurotransmitters, etc.), were likely to provoke increases in intramitochondrial free Ca 2+ ( m) concentrations. Mammalian mitochondria have a huge capacity for Ca 2+ uptake and for many years it was thought that these organelles served as mobilisable intracellular reservoirs of Ca 2+ ions.
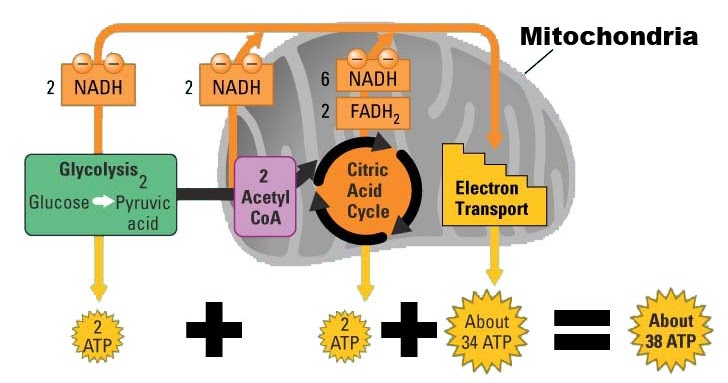
However, regulation by Ca 2+ has emerged in recent years as a further important means of controlling this vital aspect of cell function. Mitochondrial oxidative metabolism has long been recognised as subject to complex regulation by several factors, notably the concentrations of ADP and substrate(s).
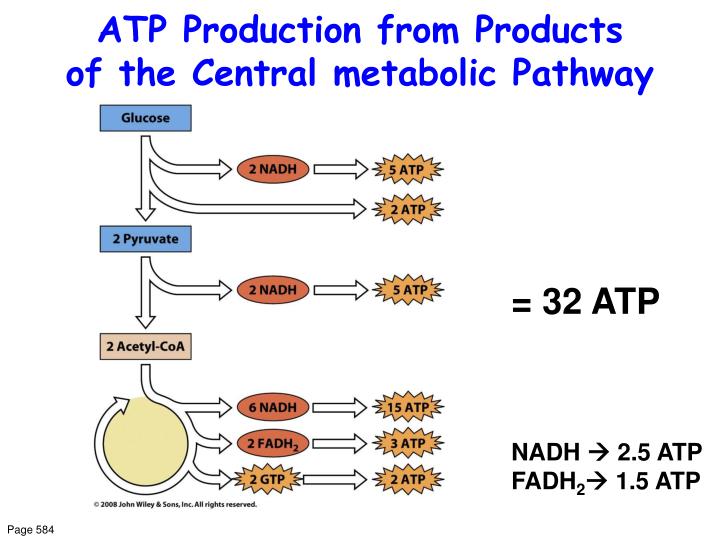
2. Ca 2+ regulation of intramitochondrial oxidative metabolism: a brief historical perspective

